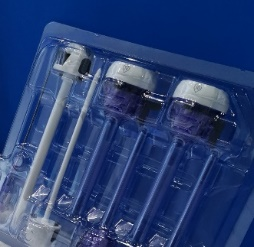
吸塑盒可用作二类或三类医疗器械的初包装材料,材质PETG或APET,EO或辐照灭菌, 大部分需要与盖材热封形成无菌屏障系统(SBS)。欧尼斯使用医疗级PETG材料(Eastar 6763或SK 2008),在万级洁净环境(ISO 7)下生产医疗吸塑盒,原材料满足生物相容性、老化、灭菌、生物负载和微粒等方面的要求。
典型应用:植入类器械、介入类器械、起搏器、外科手术包、吻合器、医美等
Blisters can be used for Class II and Class III medical devices as primary packaging. The materials are PETG or APET, EO or irradiation sterilization. Most of them need to be heat sealed with the cover material(lid) to form a Sterile Barrier System (SBS). Medical grade PETG Eastar 6763 or SK 2008 blisters are produced in Honest Packaging's Class 10000 clean rooms (ISO 7). The materials meet the requirements of biocompatibility, aging, sterilization, bioburden and particles. Typical applications: implantable devices, interventional devices, pacemakers, surgical kits, staplers, medical aesthetics, etc.